Unité de Catalyse et de Chimie du Solide site Artois
Faculté des Sciences de Lens
PUBLICATIONS MARQUANTES 2017 :
- 12 publications [à Facteur d'impact > 5]
- «Acid-tolerant cyclodextrin based ruthenium nanoparticles for the hydrogenation of unsaturated compounds in water»
S. Noël, D. Bourbiaux, N. Tabary, A. Ponchel, B. Martel, E. Monflier, B. Léger
Catal. Sci. Technol., 2017, 7, 5982-5992 - doi: 10.1039/C7CY01687E
IF = 5,365
 |
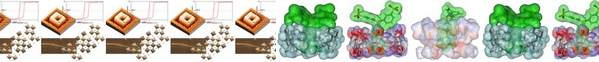
| A water-soluble β-cyclodextrin polymer synthesized by crosslinking β-cyclodextrin with epichlorohydrin and glycidyltrimethylammonium chloride allowed the stabilization of ruthenium nanoparticles in basic aqueous medium but also in acidic medium. The aqueous ruthenium colloidal suspensions obtained with this polymer were active as catalysts for the hydrogenation of a large variety of unsaturated compounds including aromatic or fatty acids. The recycling of this catalytic system was attested through ten consecutive runs without loss of stability and activity, demonstrating its robustness. |

- «Mixed Oxides supported Low-Nickel Formulations for the direct amination of aliphatic alcohols with ammonia»
A. Tomer, Z. Yan, A. Ponchel, M. Pera-Titus
J. Catal. 356 (2017) 133-146 - doi: 10.1016/j.jcat.2017.08.015
IF = 6,759
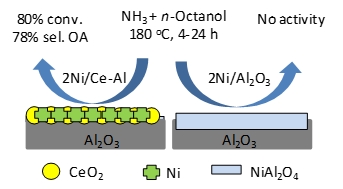 |
| The present study focuses on the selective synthesis of primary amines from aliphatic alcohols and ammonia using alumina-ceria supported nickel formulations based on very low nickel loading (≤2 wt.%) and without any external additive or H2 supply. The effect of the catalyst preparation methods and modes of nickel impregnation were studied in detail and comprehensively characterized. The best formulation afforded 80% n-octanol conversion with 78% selectivity to n-octylamine at optimized reaction conditions, which were far better than control catalysts and benchmark Ni-alumina formulations relying on high Ni loadings. The enhanced activities of the alumina-ceria supported nickel catalysts was attributed to three combined effects: (1) a higher reducibility of surface nickel oxide species, (2) the genesis of very small and homogeneously distributed nickel nanoparticles (2-3 nm), and (3) a strong decline in the formation of nickel aluminates. Furthermore, unlike benchmark Ni catalysts, these formulations afforded a higher resistance to leaching. |

- «Facile preparation of Ni/Al2O3 catalytic formulations with the aid of cyclodextrin complexes: towards highly active and robust catalysts for the direct amination of alcohols»
A. Tomer, F. Wyrwalski, C. Przybylski, J.-F. Paul, E. Monflier, M. Pera-Titus, A. Ponchel
J. Catal. 356 (2017) 111-124 - doi: 10.1016/j.jcat.2017.10.006
IF = 6,759
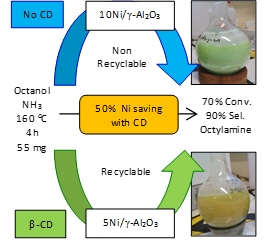 |
| A series of Ni/Al2O3 catalysts with variable Ni loading (2-20 wt.%) were prepared by aqueous wet impregnation of a nitrate precursor using native cyclodextrins as metal complex hosts. The impact of β-CD was carefully characterized at different stages of the preparation by a set of complementary techniques including TG-MS analysis, mass spectrometry, X-Ray diffraction, temperature-programmed reduction, CO pulse chemisorption, X-Ray photoelectron spectroscopy and electron microscopy. It was found that the use of cyclodextrins afforded a much higher Ni dispersion and narrower distribution of Ni particle sizes, as well as a much higher availability of reduced surface Ni species. As a result, the cyclodextrin-assisted catalysts exhibited enhanced catalytic properties in the direct amination of benzyl alcohol with aniline and n-octanol with ammonia, both operated via the hydrogen borrowing mechanism. Furthermore, the use of cyclodextrins allowed a significant improvement of the robustness of the catalysts by mitigating the nickel leaching during reaction. |

- «Nonconventional Three-Component Hierarchical Host-Guest Assembly Based on Mo-Blue Ring-Shaped Giant Anion, γ-Cyclodextrin and Dawson-type Polyoxometalate»
M. Aly Moussawi, M. Haouas, S. Floquet, W.E. Shepard, P.A. Abramov, M.N. Sokolov, V.P. Fedin, S. Cordier, A. Ponchel, E. Monflier, J. Marrot, E. Cadot
J. Am. Chem. Soc. 2017, 139, 14376-14379 - doi: 10.1021/jacs.7b08058
IF = 14,357
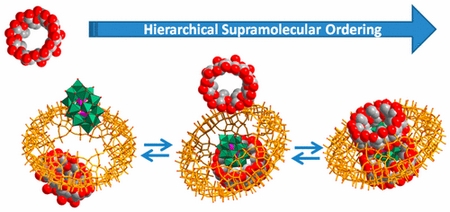 |
| In this communication, we report on a noteworthy hybrid supramolecular assembly built from three functional components hierarchically organized through noncovalent interactions. The one-pot synthesis procedure leads to the formation of large Mo-blue ring-shaped anion {Mo154}, which contains the supramolecular adduct based on the symmetric encapsulation of the Dawson-type [P2W18O62]6– anion by two γ-cyclodextrin units. Such a nanoscopic onion-like structure, noted [P2W18O62]@2γ-CD@{Mo154} has been characterized by single-crystal X-ray diffraction, thus demonstrating the capability of the giant inorganic torus to develop relevant supramolecular chemistry, probing the strong affinity of the inner and outer faces of the γ-CD for the polyoxometalate surfaces. Furthermore, interactions and behavior in solution have been studied by multinuclear NMR spectroscopy, which supports specific interactions between γ-CD and POM units. Finally, the formation of this three-component hybrid assembly from one-pot procedure, in water and using nearly stoichiometric conditions, is discussed in terms of the driving forces orchestrating this highly efficient multilevel recognition process. |

- «Polyoxometalate, cationic cluster and g-cyclodextrin. From primary interactions to supramolecular hybrid materials»
M. Aly Moussawi, N. Leclerc, P. Abramov, M.N. Sokolov, S. Cordier, A. Ponchel, E. Monflier, H. Bricout, D. Landy, M. Haouas, J. Marrot, E. Cadot
J Am Chem Soc 2017, 139, 12793-12803 - doi: 10.1021/jacs.7b07317
IF = 14,357
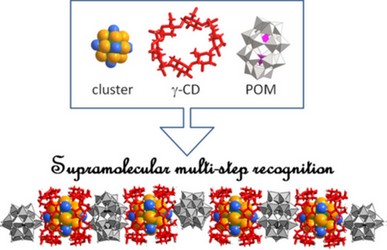 |
| Herein, we report on a three-component supramolecular hybrid system built from specific recognition processes involving a Dawson type polyoxometalate (POM), [P2W18O62]6-, a cationic electron-rich cluster [Ta6Br12(H6O)6]2+ and γ-cyclodextrin (γ-CD). Such materials have been investigated using a bottom-up approach by studying the specific interactions between γ-CD and both types of inorganic units. Their ability to interact has been investigated in the solid state by single-crystal X-ray diffraction (XRD) and in solution using multinuclear NMR methods (including DOSY, EXSY, and COSY), electrospray ionization mass and UV–vis spectroscopies, electrochemistry, and isothermal titration calorimetry experiments. Single-crystal XRD analysis reveals that POM:γ-CD constitutes a highly versatile system which gives aggregates with 1:1, 1:2, and 1:3 stoichiometry. Surprisingly, these arrangements exhibit a common feature wherein the γ-CD moiety interacts with the Dawson-type POMs through its primary face. We present also the first structural model involving an octahedral-type metallic cluster with γ-CD. XRD study reveals that the cationic [Ta6Br12(H2O)6]2+ ion is closely embedded within two γ-CD units to give a supramolecular ditopic cation, suitable to be used as a linker within extended structure. Solution study demonstrates clearly that pre-associations exist in solution for which binding constants and thermodynamic parameters have been determined, giving preliminary arguments about the chaotropic nature of the inorganic ions. Finally, both building blocks i.e. the ditopic supramolecular cation {[Ta6Br12(H6O)6]@2CD}2+ and the Dawson-type anion react together to give three-component well-ordered hybrid material derived either as supramolecular hydrogel or single-crystals. Solid state structure shows an unprecedented helicoidal tubular chain resulting from the periodic alternation of POM and supramolecular cation, featured by short hydrogen bonding contact between electron-poor POM and electron-rich cluster. The 1D tubular ionic polymer observed in the single-crystals should allow understanding the long-range ordering observed within the hydrogel hybrid material. The supramolecular chemical complementarities between the γ-CD- based ditopic cation and polyoxometalate open a wide scope for the design of hybrid materials that cumulate synergistic functionalities. |

- «Water-soluble Phosphane-Substituted Cyclodextrin as Effective Bifunctional Additive in Hydroformylation of Higher Olefins»
J. Leblond, J. Potier, S. Menuel, H. Bricout, C. Machut-Binkowski, D. Landy, S. Tilloy, E. Monflier, F. Hapiot
Catal. Sci. Technol., 2017, 7, 3823 – 3830 - doi: 10.1039/C7CY01108C
IF = 5,365
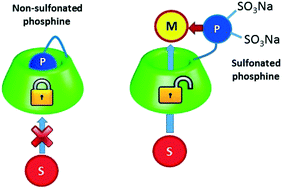 |
| In cyclodextrin (CD)-mediated aqueous biphasic catalysis, favoring contacts between the CD (“host”), the organic substrate (“guest”) and the water-soluble catalyst is crucial for the reaction to proceed efficiently at the aqueous/organic interface. Grafting the catalyst onto the CD backbone thus appears as an attractive approach to favor the molecular recognition of the substrate and its subsequent catalytic conversion into products. In this context, a new water-soluble β-CD-based phosphane was synthesized and characterized by NMR, tensiometric and ITC measurements. The β-CD-based phosphane consisted of a 3,3′-disulfonatodiphenyl phosphane connected to the primary face of β-CD by a dimethyleneamino spacer. Intra- and intermolecular inclusion processes of one of the two sulfophenyl groups into the β-CD cavity were identified in water. However, the association constant (Ka) related to the β-CD/sulfophenyl group couple was low. Accordingly, the inclusion process was easily displaced upon coordination to rhodium complexes. The efficacy of the resulting Rh-complex coordinated by β-CD-based phosphanes was assessed in Rh-catalyzed hydroformylation of higher olefins. The catalytic system proved to be far more successful and efficient than a system consisting of supramolecularly interacting phosphanes and CDs. The catalytic activity was up to 30-fold higher while the chemo- and regioselectivities remain rather unchanged. |

- «Hydroformylation of alkenes in a planetary ball mill: from additive-controlled reactivity to supramolecular control of regioselectivity»
K. Cousin, S. Menuel, E. Monflier, F. Hapiot
Angew. Chem. Int. Ed. 2017, 56, 10564-10568 - doi: 10.1002/anie.201705467
IF = 12,102
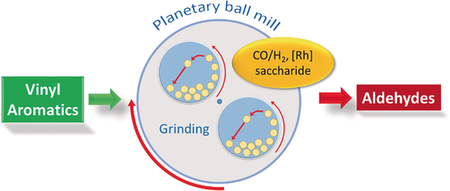 |
| The Rh-catalyzed hydroformylation of aromatic-substituted alkenes is efficiently performed in a planetary ball mill under CO/H2 pressure. The dispersion of the substrate molecules and the Rh-catalyst into the grinding jar is ensured by saccharides, namely, methyl-α-D-glucopyranoside, maltodextrins or cyclodextrins (CDs). Aldehydes were exclusively formed whatever the saccharide. Acyclic saccharides efficiently disperse the components within the solid mixture leading to high conversions of alkenes. They showed typical selectivity for α-aldehyde products. On the other hand, with CDs as dispersing additive, the steric hindrance exerted by the CDs with respect to the primary coordination sphere of the metal modifies the selectivity to such an extent that the β-aldehydes were also formed in non-negligible proportions. Such through-space control via hydrophobic effects over reactivity and regioselectivity reveals the potential of such solventless process for catalysis in solid state. |

- «Hydroaminomethylation/hydrohydroxymethylation sequence for the one pot synthesis of aminohydroxytriglycerides»
T. Vanbesien, E. Monflier, F Hapiot
Green Chem. 2017, 19, 1940-1948 - doi: 10.1039/C7GC00061H
IF = 8,586
 |
| In this paper we report the one pot functionalization of the carbon-carbon double bonds of triglycerides via a cascade hydroaminomethylation (HAM) / hydrohydroxymethylation (HHM) reaction. The carbon-carbon double bonds are first hydroformylated using a Rh-catalyst. The produced aldehydes react with secondary amines to yield enamines which are then reduced into tertiary amines. Interestingly, the latter act as ligands of Rh-species capable of hydrogenating the remaining formyl groups into alcohols. As a result, the final triglycerides are substituted by both aminomethyl and hydroxymethyl groups. Of interest, no phosphane ligand is used to stabilize the Rh-species. Moreover, the proportion of aminomethyl and hydroxymethyl groups grafted onto the triglyceride fatty chains can be finely adjusted by a careful choice of the experimental conditions, especially the nature and the amount of the amine, the reaction temperature, and the CO/H2 pressure. The resulting amino-hydroxylated triglycerides are unprecedented biosourced building blocks whose applications could be of major interest, especially in the field of polymer chemistry. |

- «Cyclodextrin-directed synthesis of gold-modified TiO2 materials and evaluation of their photocatalytic activity in the removal of a pesticide from water. Effect of porosity and particle size»
A. Lannoy, R. Bleta, C. Machut-Binkowski, A. Addad, E. Monflier, A. Ponchel
ACS Sustainable Chem. Eng. 2017, 5, 3623-3630 - doi: 10.1021/acssuschemeng.6b03059
IF = 6,140
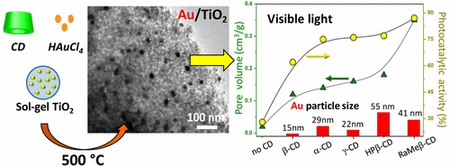 |
| Visible-light-driven plasmonic photocatalysts are promising candidates for environmental cleanup applications owing to their high solar energy utilization efficiency. In this Letter, we describe a facile and versatile cyclodextrin (CD)-driven colloidal self-assembly approach toward visible-light-responsive Au/TiO2 photocatalysts for the degradation of the herbicide phenoxyacetic acid (PAA) in water. The effect of several cyclodextrins, natives and modified, is investigated. Our results reveal that these cyclic oligosaccharides exert antagonistic actions on the size of metal particles and the porosity of composite materials. Indeed, because of their surface-active properties and weak intermolecular interactions, the randomly methylated β-CD and the 2-hydropropoxyl β-CD produce large Au particles dispersed over a highly porous TiO2 material, whereas the native α-CD, β-CD and γ-CD, owing to their ability to self-assemble into cage-type structures through intermolecular hydrogen-bond interactions, allow us to fabricate small Au particles dispersed over a dense and compact network. Among the various cyclodextrins investigated, the randomly methylated β-CD leads to Au/TiO2 composites with an appropriate combination of interconnected pore structure, large surface area, high crystallinity and optimum Au particle size for the best photocatalytic activity under visible-light irradiation. Our findings highlight the pivotal role played by the chemical nature of the substituents in the CD ring on the structural and textural characteristics of the composites as well as on their photocatalytic degradation efficiency. |

- «Catalysis in cyclodextrin-based unconventional reaction media: recent developments and future opportunities»
F. Hapiot, S. Menuel, M. Ferreira, B. Léger, H. Bricout, S. Tilloy, E. Monflier
ACS Sustainable Chem. Eng. 2017, 5, 3598-3606 - doi: 10.1021/acssuschemeng.6b02886
IF = 6,140
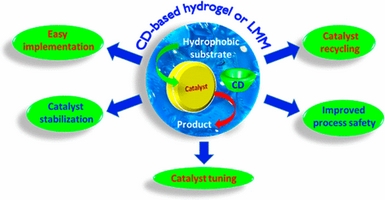 |
| During these last years, cyclodextrins (CDs) have greatly contributed to the development of innovative homogeneous or heterogeneous catalytic processes. More than simple molecular platforms aiming at designing new ligands or interfacial additives, CDs have been employed to generate unconventional reaction media such as supramolecular hydrogels or low melting mixtures (LMMs) capable of stabilizing active catalytic species. By using such alternative and unconventional media, high catalytic activities and selectivities were obtained in various transition metal catalytic reactions. The studied catalytic systems are easy to implement and allow for the remarkable stabilization of organometallic or metal nanoparticle catalyst. Interestingly, the size of metal nanoparticles can be finely tuned through confinement in the network of CD-based supramolecular hydrogels. Additionally, the catalysts can be recovered and recycled quantitatively by a simple phase separation. The catalytic phase can be stored at room temperature under air-atmosphere in the solid state and reused as needed without significant loss of activity. Eventually, such CD-based catalytic systems greatly improve the process safety. The present article intends to show the reader the very substantial progress that has recently been made in the field. |

- «Reduction of Ln2Ti2O7 Layered Perovskites: A Survey of the Anionic Lattice, Electronic Features, and Potentials»
T. Pussacq, H. Kabbour, S. Colis, H. Vezin, S. Saitzek, O. Gardoll, C. Tassel, H. Kageyama, C. Laberty Robert, O. Mentré
Chem. Mater., 2017, 29 (3), pp 1047–1057 - doi: 10.1021/acs.chemmater.6b03808
IF = 9,890
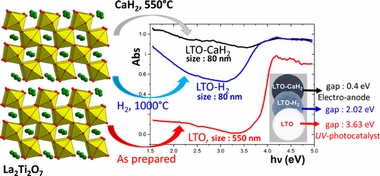 |
| The reduction of the layered perovskites Ln2Ti2O7 (LnTO, with lanthanide Ln = La, Pr, or Nd) was studied with the aim of shifting the ultraviolet (UV) photocalytic activity for water splitting in the visible range by Ti3+ donor doping. For all phases, after reduction by CaH2, the absorbance is extended beyond the UV–visible region, giving rise to a gaplike edge in the mid-infrared at ∼0.4 eV with a dark coloration of the samples. When the precursor with Ln = La was reduced under a high-temperature H2 flow, we found a progressive nanotexturation down to 300 nm, which is responsible for a degree of Ti3+ segregation at the surface. Magnetic measurements, thermal analysis, and powder neutron diffraction reveal that the samples reduced by both routes have a similar amount of anion vacancy with δ = 0.27 (in La2Ti2O7−δ). It represents a limited topotactic reduction stage, prior to the reconstructive reduction into La5Ti3.8+5O17 observed under more severe reducing conditions. For the sample reduced by CaH2, a minor amount of hydride appears to be incorporated (∼0.02 H per FU), with Ti3+···H– bonding observed by hyperfine sublevel correlation spectroscopy electron paramagnetic resonance and density functional theory calculations. Preliminary electrocatalysis tests show a promising anodic activity for water splitting hydrogen evolution with a voltage onset as low as 0.6 V versus the reversible hydrogen electrode. |

- «Tetronics/cyclodextrin-based hydrogels as catalyst-containing media for the hydroformylation of higher olefins»
M. Chevry, T. Vanbesien, S. Menuel, E. Monflier, F Hapiot
Catal. Sci. Technol., 2017, 7, 114-123 - doi: 10.1039/C6CY02070D
IF = 5,365
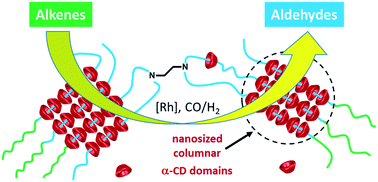 |
| The rhodium-catalyzed hydroformylation of alkenes has been investigated in biphasic conditions using combinations of α-cyclodextrin (α-CD) and poloxamines (Tetronics®). Thermo-responsive hydrogels containing the Rh-catalyst are formed under well-defined conditions of concentration. Hydrogels consisting in the reverse-sequential Tetronic®90R4 prove to be more effective than the conventional sequential Tetronics®701. The presence of α-CD is crucial to provoke the decantation of the multiphasic system once the reaction is complete. Optimized conditions (CO/H2 pressure, Rh-precursors, phosphanes…) show that the catalytic system is especially applicable to the hydroformylation of terminal alkenes. The catalytic performance remain unchanged upon recycling as the hydrogel matrix prevents the oxidation of the phosphane. |

Faculté Jean Perrin - rue Jean Souvraz - SP 18 - 62307 Lens Cedex
tel : 03 21 79 17 05
fax : 03 21 79 17 55 |