Unité de Catalyse et de Chimie du Solide site Artois
Faculté des Sciences de Lens
PUBLICATIONS MARQUANTES 2013 :
- 7 publications [à Facteur d'impact > 5]
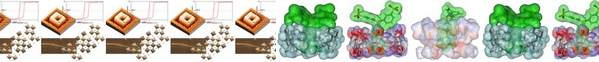
- «A Cyclodextrin Dimer as Supramolecular Reaction Platform for Aqueous Organometallic Catalysis»
C. Blaszkiewicz, H. Bricout, E. Léonard, C. Len, D. Landy, C. Cézard, F. Djedaïni-Pilard, E. Monflier, S. Tilloy
Chem. Commun. 2013, 49, 6989-6991 - doi: 10.1039/C3CC43647K
IF = 6,718
 |
| A reaction platform based on a cyclodextrin dimer, which is able to simultaneously include a substrate in one cavity and an organometallic catalyst into the other, proved to be highly efficient for aqueous hydroformylation reaction of higher olefins. |

- «Co-Assembly of Block Copolymer and Randomly-Methylated β-Cyclodextrin: From Swollen Micelles to Mesoporous Alumina with Tunable Pore Size»
R. Bleta, C. Machut, B. Léger, E. Monflier, A. Ponchel
Macromolecules 2013, 46 (14), pp 5672–5683 - doi: 10.1021/ma4008303
IF = 5,927
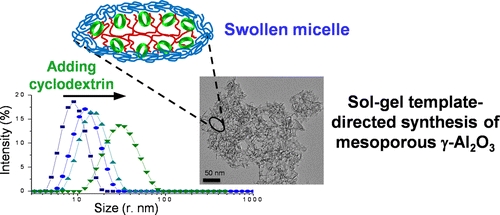 |
| Controlling interactions at the supramolecular level is of importance for the preparation by template directed synthesis of mesoporous materials with tailored pore structures. Herein we investigate the effect of randomly methylated β-cyclodextrin on the association behavior of the amphiphilic triblock copolymer Pluronic P123 in aqueous solution. Surface tension, dynamic light scattering and viscosity measurements provide quantitative evidence that, when the cyclodextrin is added in controlled amounts to the copolymer, it strongly impacts the micellar growth rate. The property of randomly methylated β-cyclodextrin to act as a micelle expander has been further exploited to generate a series of mesoporous -alumina by a sol-gel method. The resulting materials subjected to calcination at 500 °C exhibit high surface area (354 to 382 m2/g), tunable pore size (14.8 to 19.3 nm) and very large pore volume (1.37 to 1.97 cm3/g), making them excellent candidates for applications in the fields of adsorption and heterogeneous catalysis. |

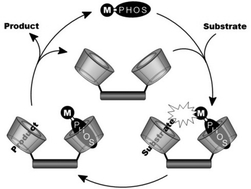
- «Effects of ß-cyclodextrin introduction to zirconia supported-cobalt oxide catalysts: from molecule-ion associations to complete oxidation of formaldehyde»
L. Bai, F. Wyrwalski, J-F. Lamonier, A.Y. Khodakov, E. Monflier, A. Ponchel
Appl. Catal. B: Environ., 2013, vol.138– 139, 381– 390 - doi : 10.1016/j.apcatb.2013.03.015
IF = 6,007
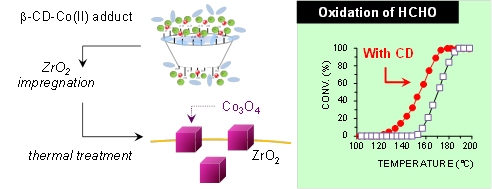 |
| The present paper deals with the impact of the addition of β-cyclodextrin on the genesis of cobalt active phases in zirconia-supported catalysts and on their performances in the complete oxidation of formaldehyde. With this aim, a series of cobalt catalysts supported on zirconia (5 wt.%) is prepared by wet impregnation using mixtures of Co(NO3)2 and β-cyclodextrin as precursors. After the impregnation step, the materials are dried and calcined under air at 400 °C. Different ratios of β-cyclodextrin to cobalt have been employed to obtain insight in the role of β-cyclodextrin, i.e. from 0.01 to 1. The ability of β-cyclodextrin to interact with the cobalt salt is investigated in solution by UV–vis spectroscopy, ion conductivity in water and dynamic light scattering measurements. After the impregnation step, the cobalt supported materials are characterized at different stages of the preparation (before and after calcination) by means of conventional techniques including UV–vis spectroscopy, thermogravimetry, N2 adsorption/desorption, X-ray diffraction and temperature-programmed reduction analysis. Characterization results suggest that the carbohydrate ligand bound to the cobalt species retards the decomposition of the cobalt complexes into Co3O4. Finally, these cobalt oxide catalysts prepared from β-cyclodextrin are able to catalyze the oxidation of formaldehyde more efficiently than the control sample. The results are explained in terms of cobalt oxide dispersion and cobalt-support interactions. Indeed, the cobalt oxide particles generated by the thermal decomposition of β-cyclodextrin-Co(II) adducts are on the one hand smaller than those formed with cobalt nitrate alone and on the other hand reducible at lower temperatures, two key factors in the catalytic oxidation reactions. |

- «Rhodium-Catalyzed Homogeneous and Aqueous Biphasic Hydroformylation of the Acrolein Acetal 2-Vinyl-5-Methyl-1,3-Dioxane»
J. Ternel, J.L. Dubois, J.L. Couturier, E. Monflier, J.F. Carpentier
ChemCatChem, Volume 5, Issue 6, pages 1562–1569, June 2013 - doi: 10.1002/cctc.201200630
IF = 5,044
| The rhodium-catalyzed hydroformylation of 2-vinyl-5-methyl-1,3-dioxane (VMD), the acetal derived from acrolein and 2-methyl-1,3-propanediol, has been investigated. Under homogeneous conditions, phosphane-based catalysts afforded perfect chemoselectivity for the aldehydes. Catalysts based on diphosphine ligands with a narrow bite angle of approximately 85°, such as 1,2-bis(phenylphosphino)ethane, were found to favor the branched aldehyde 2, with selectivities of up to 78 %, whereas diphosphine ligands with a large bite angle of approximately 110°, such as xantphos, produce the linear aldehyde 1 with up to 91 % selectivity. Control of the 1/2 regioselectivity by means of the P=Rh=P bite angle was ascertained with a variety of ethylene-, hydrazino- and CNC-bridged diphosphines, irrespective of electronic factors induced by these ligands. Under aqueous biphasic conditions, chemoselective hydroformylation of VMD proved feasible only with disulfonated xantphos; in this case, remarkable activities (turnover frequencies of up to 1075 molaldehyde molRh−1 h−1 at 120 °C) were observed, with up to 82 % yield for the linear aldehyde. On the other hand, the catalyst based on tetrasulfonated dppe systematically induced the formation of very large amounts of the hydrogenated product. |

- «Methylated β-Cyclodextrin-Capped Ruthenium Nanoparticles: Synthesis Strategies, Characterization, and Application in Hydrogenation Reactions»
N. T. T. Chau, S. Handjani, J.-P. Guegan, M. Guerrero, E. Monflier, K. Philippot, A. Denicourt-Nowicki, A. Roucoux
ChemCatChem, Volume 5, Issue 6, pages 1497–1503, June 2013 - doi: 10.1002/cctc.201200718
IF = 5,044
| Colloidal suspensions of ruthenium nanoparticles (Ru0 NPs) stabilized by randomly methylated β-cyclodextrins were easily prepared in aqueous solution by two comparative reductive approaches. The first method relies on a one-step hydrogen reduction of ruthenium trichloride as a metal precursor in the presence of cyclodextrins (one-pot method). The second method consists of the chemical reduction of metal salts by NaBH4, followed by the stabilization of ruthenium NPs by post-addition of the methylated β-cyclodextrins (cascade method). The modified β-cyclodextrin-capped ruthenium NPs obtained were characterized using TEM, DLS, and NMR techniques. The highly dispersed colloidal suspensions contain very small particles in the size range of 1–1.4 nm. Their catalytic performances were investigated in terms of activity and selectivity of their biphasic hydrogenation of various substrates (olefins, ketones, and disubstituted arenes) under mild conditions. |

- «Thermoresponsive Hydrogels in Catalysis»
F. Hapiot, S. Menuel, E. Monflier
ACS Catalysis 2013, 3(5), 1006-1010 - doi: 10.1021/cs400118c
IF = 7,572
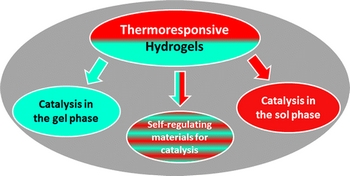 |
| The tunable properties of thermoresponsive physical hydrogels recently found application in catalysis. The most relevant examples are described in this perspective article. Novel concepts are especially highlighted through the beneficial effects of thermoresponsive hydrogels on the catalytic performance. Their scope and future developments are also addressed. |

- «Pickering Emulsions Based on Supramolecular Hydrogels: Application to Higher Olefins’ Hydroformylation»
J. Potier, S. Menuel, M.-H. Chambrier, L. Burylo, J.-F. Blach, P. Woisel, E. Monflier, F. Hapiot
ACS Catalysis 2013 , 3, pp 1618–1621 - doi: 10.1021/cs4002282
IF = 7,572
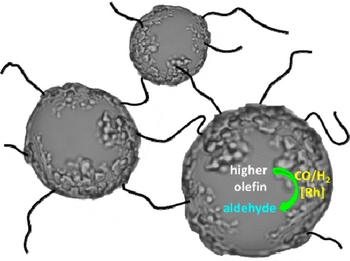 |
| Supramolecular hydrogels elaborated from a mixture of native α-cyclodextrin and poly(ethylene glycol)s in water proved to be effective media for higher olefins Rh-catalyzed hydroformylation due to the formation of Pickering emulsions. |

Faculté Jean Perrin - rue Jean Souvraz - SP 18 - 62307 Lens Cedex
tel : 03 21 79 17 05
fax : 03 21 79 17 55 |