Unité de Catalyse et de Chimie du Solide site Artois
Faculté des Sciences de Lens
PUBLICATIONS MARQUANTES 2021 :
- «Cyclodextrins as multitask agents for metal nano-heterogeneous catalysis: a review»
S. Noël, B. Léger, A. Ponchel, S. Sadjadi, E. Monflier
Environ Chem Lett 19, 4327–4348 (2021) -
doi: 10.1007/s10311-021-01298-5
IF = 9,027
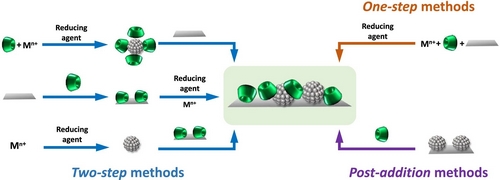 |
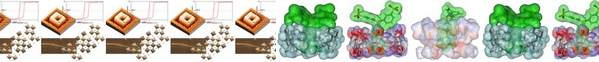
| Research in green chemistry has recently focussed on designing reactions in water as a safe solvent, calling for catalysts that operate in aqueous solutions. In particular, supramolecular metal nanoparticles have been synthesized by associating macrocyclic compounds with transition metal nanoparticles, to produce water-dispersible catalytic systems of high performance and molecular recognition ability. Here we review methods for stabilizing catalytically active metal nanoparticles in cyclodextrin-based systems. Indeed, cyclodextrins form inclusion complexes with molecules of appropriate size and shape, and the presence of hydroxyl groups enables coordination with metal ions. Representative examples show increasing stability, recyclability and catalytic activity. We present 1) the synthesis, characterization and catalytic behaviors with focus on the development of metal nanoparticles in solvent-dispersed form or immobilized onto supports in the presence of cyclodextrins or their derivatives; 2) the multi-functional role of cyclodextrins capable of acting as reducing agents, stabilizing/dispersing agents or mass transfer promoters; and 3) the design of more sophisticated catalytic systems, in which the cyclodextrin is a supramolecular host with dynamic equilibrium. |

- «Hydrohydroxymethylation of Ethyl Ricinoleate and Castor Oil»
C. Becquet, F. Berche, H. Bricout, E. Monflier, S. Tilloy
ACS Sustain Chem Eng, 2021, 9, 9444−9454 -
doi: 10.1021/acssuschemeng.1c02924
IF = 8,198
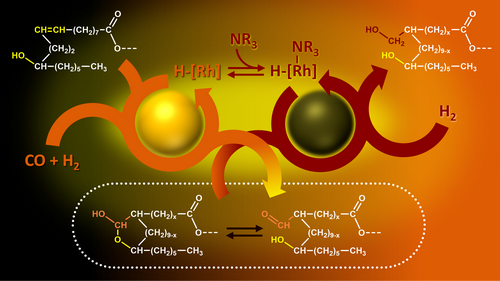 |
| The direct functionalization of the carbon–carbon double bonds of castor oil and its derivatives is of major interest to access biosourced building blocks. In particular, polyol derivatives can be produced in this way and find application in the field of bio-based polyesters and polyurethanes. In this study, we described the synthesis of polyhydroxylated derivatives via a hydrohydroxymethylation reaction consisting of two consecutive Rh-catalyzed reactions: a hydroformylation reaction followed by a hydrogenation reaction of formyl groups. A catalytic system based on Rh(acac)(CO)2 and a trialkylamine proved to be active both in hydroformylation of carbon–carbon double bonds and reduction of the resulting aldehydes into primary alcohols. By optimizing the reaction conditions, yields in alcohols of 74 and 80% were reached for castor oil and ethyl ricinoleate, respectively |

- «Oxidation of 2,5-Diformfylfuran to 2,5-Furandicarboxylic Acid Catalyzed by Candida antarctica Lipase Β Immobilized in a Cyclodextrin-Templated Mesoporous Silica. The Critical Role of Pore Characteristics on the Catalytic Performance»
C. Decarpigny, R. Bleta, A. Ponchel, E. Monflier
Colloid Surface B, 2021, 200, 111606 - doi: 10.1016/j.colsurfb.2021.111606
IF = 5,268
 |
Hypothesis
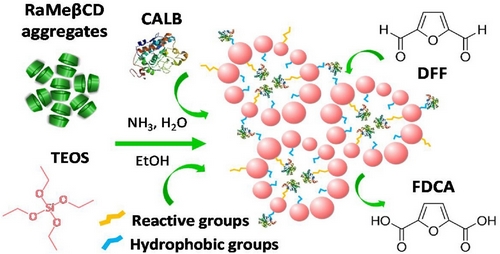
Porous silica has been extensively used as suitable carrier for the immobilization of various enzymes. Randomly Methylated β-Cyclodextrin (RaMeβCD) has surface active properties and very high solubility in water and could therefore be used as template in the fabrication of silica particles with tunable pore size.
Experiments
Silica particles were prepared by sol-gel process in alkaline medium with and without use of RaMeβCD. Lipase Bfrom Candida antarctica (CALB) was either incorporated within the pores of RaMeβCD-derived support or covalently attached on the surface of CD-free silica particles and its catalytic performance was assayed in the oxidation of 2,5-diformylfuran (DFF) to 2,5-furandicarboxylic acid (FDCA). Enzymatic reactors were characterized by N2-adsorption analysis, small angle XRD, TG/DSC experiments, ATR-FTIR spectroscopy, HR-TEM and LSCM, while reaction products were determined based on 1H NMR spectroscopy combined with HPLC.
Findings
Results showed that the use of RaMeβCD as structure directing agent led to mesoporous silica composed of uniform 8 nm-sized particles with 11 nm-sized mesopores compatible with the dimensions of CALB (3.0 nm × 4.0 nm × 5.0 nm). Incorporation of CALB within the pores of RaMeβCD-derived silica caused almost a two-fold increase in specific activity after 7 h at 40 °C when compared to lipase immobilized on the surface of CD-free silica particles (33.2 µmol g-1 min-1vs. 14.4 µmol g-1 min-1). Moreover, the RaMeβCD-derived biocatalyst demonstrated enhanced operational stability during the recycling experiments, retaining more than 90% of its initial activity after five 24 h-reaction cycles. These findings open up new avenues for future research on the use of cyclodextrins in the development of enzyme-based nanoreactors. |

- «Robust Ruthenium Catalysts Supported on Mesoporous Cyclodextrin-Templated TiO2-SiO2 Mixed Oxides for the Hydrogenation of Levulinic Acid to γ-Valerolactone»
C. Decarpigny, S. Noël, A. Addad, A. Ponchel, E. Monflier, R. Bleta
Int. J. Mol. Sci. 2021, 22, 1721.- doi: 10.3390/ijms22041721
IF = 5,923
 |
| In this paper, we present a versatile template-directed colloidal self-assembly method for the fabrication in aqueous phase of composition-tuned mesoporous RuO2@TiO2-SiO2 catalysts. Randomly methylated β-cyclodextrin/Pluronic F127 supramolecular assemblies were used as soft templates, TiO2 colloids as building blocks, and tetraethyl orthosilicate as a silica source. Catalysts were characterized at different stages of their synthesis using dynamic light scattering, N2-adsorption analysis, powder X-ray diffraction, temperature programmed reduction, high-resolution transmission electron microscopy, high-angle annular bright-field and dark-field scanning transmission electron microscopy, together with EDS elemental mapping. Results revealed that both the supramolecular template and the silica loading had a strong impact on the pore characteristics and crystalline structure of the mixed oxides, as well as on the morphology of the RuO2 nanocrystals. Their catalytic performance was then evaluated in the aqueous phase hydrogenation of levulinic acid (LA) to γ-valerolactone (GVL) under mild conditions (50 °C, 50 bar H2). Results showed that the cyclodextrin-derived catalyst displayed almost quantitative LA conversion and 99% GVL yield in less than one hour. Moreover, this catalyst could be reused at least five times without loss of activity. This work offers an effective approach to the utilization of cyclodextrins for engineering the surface morphology of Ru nanocrystals and pore characteristics of TiO2-based materials for catalytic applications in hydrogenation reactions. |

- «S=1/2 chain in BiVO3F: spin-dimers versus photoanodic properties»
O. Mentré, M. A. Juárez-Rosete, S. Saitzek, C. Aguilar-Maldonado, M. Colmont, Á. M. Arévalo-López
J AM CHEM SOC, 2021, 143, 18, 6942–6951 - doi: 10.1021/jacs.1c00621
IF = 15,419
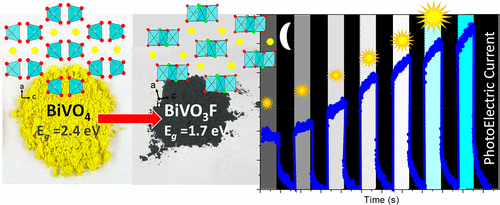 |
| BiVO3F was prepared, characterized, and identified as a unique example of bismuth vanadyl oxyhalide with paramagnetic V4+ centers. Its crystal structure shows 1D magnetic units with rare alternation of edge-sharing O–O and F–F μ2 bridges along the octahedral chains. Structural pairing across the O2 edges induces antiferromagnetic spin dimers (S = 0) with J/Kb ≈ 300 K, ∼15 times greater than the exchange across the F2 bridges, within a non-ordered magnetic ground state. Despite multiple compositional, structural, and electronic analogies with the BiVO4 scheelite compound, one of the most promising photoanodes for solar water splitting, the photoactivity of BiVO3F is relatively modest, partially due to this electronic pairing benefitting fast electron–hole recombination. Similar to monoclinic VO2, the V4+ spin dimerization deters the singlet → triplet electronic photoexcitation, but results in potential carrier lifetime benefits. The reduction of the bandgap from an Eg of ∼2.4 eV to ∼1.7 eV after incorporation of d1 cations in BiVO4 makes BiVO3F an inspiring compound for local modifications toward an enhanced photoactive material. The direct d → d transition provides a significant enhancement of the visible light capture range and opens a prospective route for the chemical design of performant photoanodes with a mixed anionic sublattice. |

- «Aqueous biphasic hydroaminomethylation enabled by methylated cyclodextrins: Sensitivity analysis for transfer into a continuous process»
K. Künnemann, D. Weber, C. Becquet, S. Tilloy, E. Monflier, T. Seidensticker, D. Vogt
ACS Sustain. Chem. Eng. 2021, 9, 273-283. - doi: 10.1021/acssuschemeng.0c07125
IF = 8,198
 |
| Hydroaminomethylation (HAM) is a highly efficient homogeneously catalyzed autotandem reaction converting alkenes into valuable amine products with water being the only coproduct. This work reports for the first time the scale-up of the highly regioselective HAM of 1-decene with diethylamine for a potential continuous process application in a green and sustainable aqueous biphasic medium. The catalytic system Rh/SulfoXantphos and randomly methylated β-cyclodextrin (RAME-β-CD) were herein dissolved in the plain aqueous phase. The addition of cyclodextrins as a green mass transfer agent remarkably increases the reaction rate as well as the selectivity toward linear amines, but they can also support solid content precipitation in this reaction system due to up-scaling effects. Therefore, parameters such as catalyst concentration, organic volume fraction, cyclodextrin concentration, and recyclability were investigated regarding the stability and activity of the system. Especially the organic volume fraction had a decisive influence on solid content precipitation. High regio- and chemoselectivities of 35 and 82%, respectively, at nearly full conversion were achieved toward the linear product amine. The catalytic system was further recycled in batch and scaled up into a 2100 mL autoclave without loss in activity and selectivity. Finally, a continuous process concept was proposed for an extensive investigation regarding long-term stability of the catalytic system. |

Faculté Jean Perrin - rue Jean Souvraz - SP 18 - 62307 Lens Cedex
tel : 03 21 79 17 05
fax : 03 21 79 17 55 |